Your Questions, Answered
We’ve assembled the most frequently asked questions about Eversense into one place. Got a question that you can’t find the answer to? Contact us.
The Eversense® E3 Sensor Kit includes:
- Pouch with the Sensor inside of a sensor holder
The Eversense® E3 Smart Transmitter Kit includes:
- Smart Transmitter
- Charging Accessory Pack
- User Guide
- Quick Reference Guide
- Wallet Card
- Next Steps Sheet
The Eversense® E3 Adhesive Patch Kit
- 150 White Adhesives
- 30 Clear Adhesives
The system is indicated (approved) for use to replace fingerstick blood glucose measurements for diabetes treatment decisions.
Pay close attention to your glucose values, symptoms, and trends. If your symptoms are different than the sensor glucose values or what the alert indicates, confirm your glucose value with a blood glucose meter test before making a treatment decision.*
To make a treatment decision, the user should consider:
- Status bar information.
- Current sensor glucose value – the current glucose value should be displayed in black.
- Trend arrow – a trend arrow should be displayed.
- Recent trend information and alerts.
When to NOT make a treatment decision:
- No glucose value is displayed.
- No trend arrow is displayed.
- Your symptoms do not match the glucose information displayed.
- The current sensor glucose value is displayed in grey.
- The status bar is displayed in orange.
- You are taking medications of the tetracycline class.

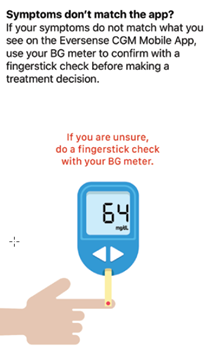

* If you have any uncertainly in how or when to make treatment decisions, always speak with your healthcare professional.
Note: Always refer to the glucose information on your Eversense CGM App on your smartphone to make treatment decisions. Do not use a secondary display like the Apple Watch or Eversense NOW.
A blood glucose meter is required for calibrating the CGM System, and to make treatment decisions under certain conditions. See Understanding Treatment Decisions with CGM in the Eversense® E3 CGM User Guide for more information.
Calibration is used to ensure the accuracy of the CGM system.
Pay close attention to your glucose values, symptoms, and trends. If your symptoms are different than the sensor glucose values or what the alert indicates, confirm your glucose value with a blood glucose meter test before making a treatment decision.
Eversense E3 has demonstrated excellent accuracy when you need it most - we’ve demonstrated 99% of high (180 mg/dL) and 94% of low (70 mg/dL) glucose events1 detected correctly and essentially no compression lows.2
1. Senseonics. (2023) Eversense E3 Continuous Glucose Monitoring System User Guide. LBL-6002-01-001_Rev C
2. Christiansen MP et al. A Prospective Multicenter Evaluation of the Accuracy of a Novel Implanted Continuous Glucose Sensor: PRECISE II. DIABETES TECHNOLOGY & THERAPEUTICS 2018; 20(3):197-206
Still have questions?
The Eversense team is dedicated and ready to provide the answers and support you need. Simply click the link below.
The Eversense® E3 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to 180 days in persons with diabetes age 18 and older. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are still required for calibration primarily one time a day after day 21, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense E3 CGM System is a prescription device; patients should talk to their health care provider to learn more.
The Eversense® 365 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to one year in people (18 years and older) with diabetes. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are required for calibration one time a week after day 13, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense 365 CGM System is a prescription device; patients should talk to their health care provider to learn more.
For important safety information, see bit.ly/eversensesafety
Eversense, Eversense E3 Continuous Glucose Monitoring, Eversense 365 Continuous Glucose Monitoring, and the Eversense logo are trademarks of Senseonics, Incorporated. Ascensia, the Ascensia Diabetes Care logo are trademarks and/or registered trademarks of Ascensia Diabetes Care Holdings AG. All other trademarks are properties of their respective owners and are used solely for informative purposes. No relationship or endorsement should be inferred or implied.
Apple Watch® is a product of Apple, Inc., and may be separately purchased from an authorized Apple retailer. Apple Watch is not included with the Eversense CGM System. Android is a trademark of Google LLC.
PP-SENS-E3-GBL-0060